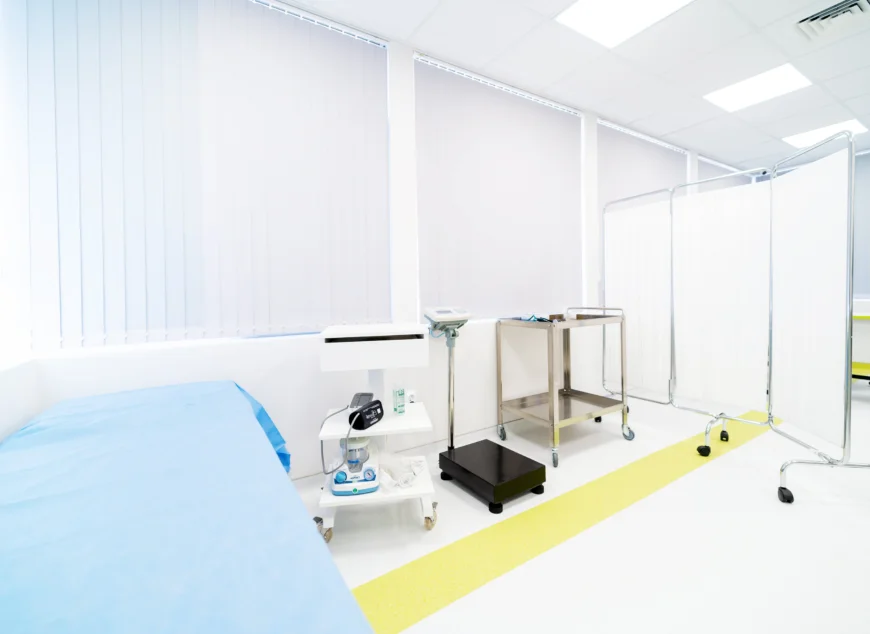
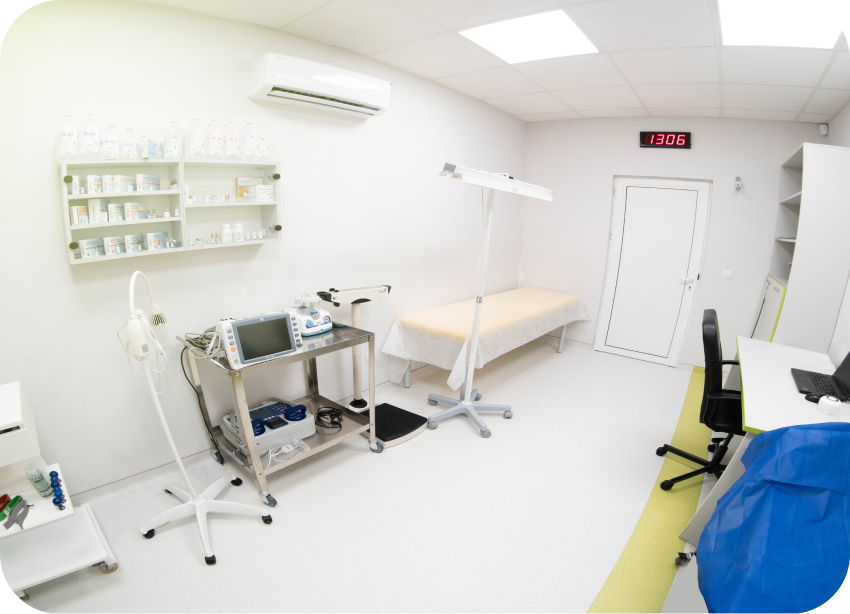
Introducing Comac Medical’s Early Phase Clinical Research Unit (CRU), where over 18 years of expertise in Phase I, Bioavailability and Bioequivalence (BA/BE) studies meet state-of-the-art facilities. Strategically located in Sofia, Bulgaria, our Phase I Unit is the finest in Europe and largest in Southeastern Europe.
Strategic Advantages of Our Phase I Clinical Trial Unit
Expertise & Specialization
Accelerated Timelines
Rapid Patient Enrollment
Collaborative Support
Independent Structure
Parallel Conduct of Trials
Key Features of Our Early Phase Clinical Research Unit
1200 square meters
(Including Intensive Care Ones)
Medical Team
Team & Equipment
Cohorts / Trials
Clinical Team
Dedicated Pharmacy
Aseptic Medication Preparation
Sampling Room
Surveillance
Alarm Systems
Recording Systems